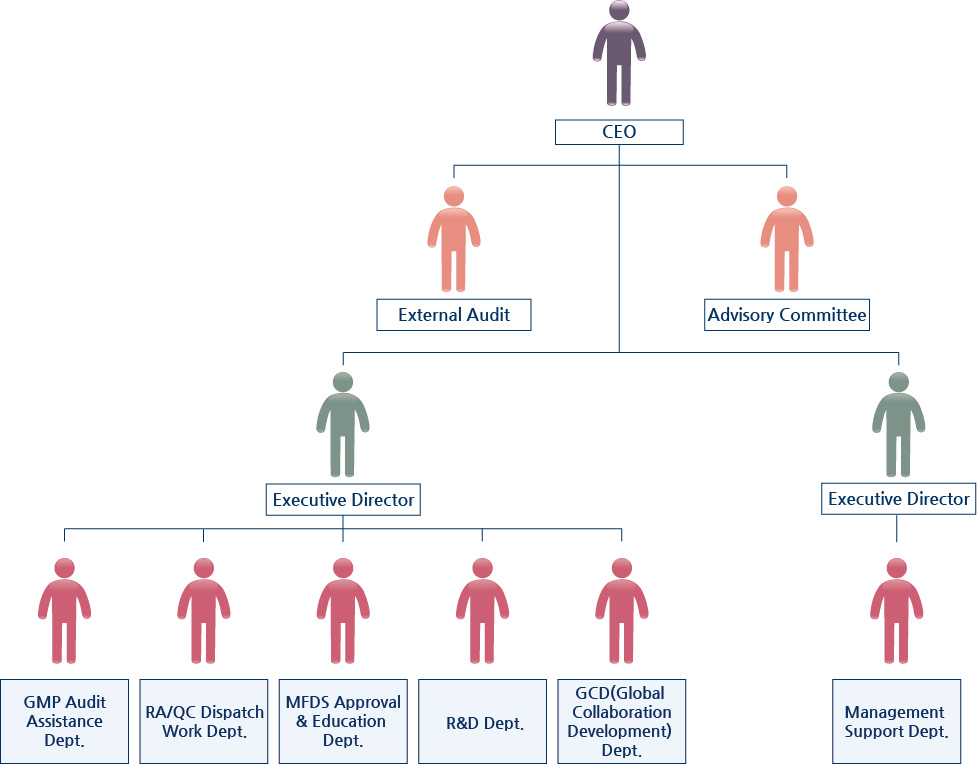
CEO's Message

Korea Medical Industry Institute of Goods Management Procedure (K-GMP) aims to provide accurate and prompt advice on MFDS in regards to Domestic and imported medical devices and to offer objective and reliable information about health and medical technology through a colossal amount of database.
In addition, based on valuable experience and competence, K-GMP wishes to serve as the most reliable assistant in helping you to become the forefront competitor in a market with strengthening regulations according to countries and dynamic conditions. We offer you solutions for your business with our deep expertise in regulations and reimbursement system necessary for manufacturing, importing and sales of medical device, food, cosmetic and drug.
Also, we will always try to achieve what we promise with our clients as a representing example of Korea female-owned business.
Many thanks.
| CEO |
 |
| Christine Kim |
History
 2012
2012
| - |
Foundation of K-GMP Co., Ltd |
2013
| - |
Taiwan Gale Med GMP Audit Interpretation |
| - |
Germany Surgiceye Audit GMP Interpretation |
| - |
France Tornier GMP Audit Interpretation |
| - |
Korea Agro_Fisheries & Food Trade Corp. U.S. FDA Conference |
| - |
MOU with Registrar Corp. |
| - |
Singapore Health Status GMP Audit Interpretation |
| - |
Domestic Medical Device Hospital Facility Exhibition |
| - |
Italy K-Laser GMP Audit Interpretation |
| - |
Certificate of Patent Application (Business Name) |
| - |
BMCC Marketing Strategy Conference |
| - |
Finland INION GMP Audit Interpretation |
2014
| - |
SPC Paris croissant O-water has been registered in FDA |
| - |
MOU with CE.way & 北京采瑞医药科技有限公司 |
| - |
KOREA EUNDAN Solar- C has been registered in CFDA |
| - |
The Truemetic ISO22716 CE Audit Interpretation |
| - |
Chunho FOOD FDA Audit Interpretation |
| - |
SEOUL FOOD(2014), Global food Standard Conferenc |
| - |
SOLCO BIO MEDICAL FDA 510K submitted |
| - |
FDA/CFDA Cosmetics & Food Approval Seminar |
| - |
Certificate of Women Enterprise |
K-GMP was established on Nov 1,2012 to facilitate the quality management system and regulatory affairs for medical devices manufacturing/exporting companies, and to become an professional education institute for GMP audit as GMP audit agent.
We have provided our expertise for Food, Drugs, Cosmetics and Medical device companies about FDA compliance since we had MOU with U.S biggest FDA Agent, Registrar Corp.